Yeast cell wall and the innate immune system response
Melina Bonato and Liliana Borges (R&D, ICC Brazil)
Poultry production systems are highly challenging. They are affected by environmental factors like temperature, ventilation, population density and other stressing agents (Rocha et al., 2014). Additionally, pathogens and mycotoxins are constantly present, which may reduce or affect the immune response of broilers.
Broilers have a large amount of lymphoid tissue and immune system cells in the intestinal mucosa, which is known as GALT (gut-associated lymphoid tissue) and forms MALT (mucosa-associated lymphoid tissue). GALT is continuously exposed to food antigens, microflora, and pathogens (Dalloul & Lillehoj, 2006), and its role is to identify components in the intestinal lumen that can be a potential threat to the animal.
The first line of immune system defense is formed by phagocytes; this includes macrophages, heterophils (equivalent to neutrophils in mammals), dendritic cells, and natural killer cells (Sharma, 2003). Toll-like receptors, located on the surface of these immune cells, recognize microbial patterns and induce an immediate innate immune response. After activation and phagocytosis, phagocytes (“APC” antigen-presenting cells) bear an antigen processed fragment, and a chain response is initiated against it. Pathogen recognition by the innate immune system triggers immediate innate defenses and, subsequently, activates an adaptative immune response (Lee & Iwasaki, 2007).
It is important to point out that these innate immune system responses require a large number of nutrients and, especially, metabolism energy, as it is an unspecific and pro-inflammatory response, but necessary to control proliferation, invasion, and damages caused by the antigen in the animal. However, an extended pro-inflammatory response can lead to secondary diseases, immunosuppression, maintenance of immune homeostasis, intestinal dysbiosis and, finally, reduced performance and mortality.
In addition to modulating different physiological processes, including nutrition, metabolism and pathogen elimination, the intestinal microbiota of animals can change the physiopathology of parasitic diseases, conferring resistance or promoting enteric parasitic infections and playing an important role in regulating immune system response. Bacteria naturally found in the gut act as molecular adjuvants, providing indirect immunostimulation, helping the body protect itself from infections.
Therefore, an effective immunization program, a balanced diet, reduced stress factors, good management practices, and animal welfare can considerably reduce the incidence of immunosuppression. However, some feed additives can help modulate the innate immune system and, therefore, improve response to challenges.
Saccharomyces cerevisiae (ImmunoWall®, ICC Brazil) yeast cell wall, originated from the sugarcane fermentation that produces ethanol, contains approximately 55% of total indigestible carbohydrates; 35% of them are β-glucans (β-1.3 and β-1.6), and 20% of them are mannan oligosaccharides (MOS). β-glucans are recognized by phagocytes cells (Petravić-Tominac et al., 2010), which trigger an innate response that helps the animal modulate its immune system. MOS can agglutinate pathogens that have type 1 fimbriae, such as several strains of Salmonella and Escherichia coli.
A recent study by Beirão et al. (2018), where broilers were given ImmunoWall® (0.5 kg/ton) and infected at 2 days old with Salmonella Enteritidis [SE] (orally at 108 CFU/broiler), showed that at 4 and 8 days old (two and six days after infection, respectively) ImmunoWall® had reduced marker passage (Dextran-FITC, 3-5 kD) to blood in challenged broilers. These results show a significant improvement in intestinal permeability, as SE can stick to the mucosa through its fimbriae, produce toxins and damage tight junctions and enterocytes, invading them and moving to the bloodstream and other organs and tissues (Figure 1).
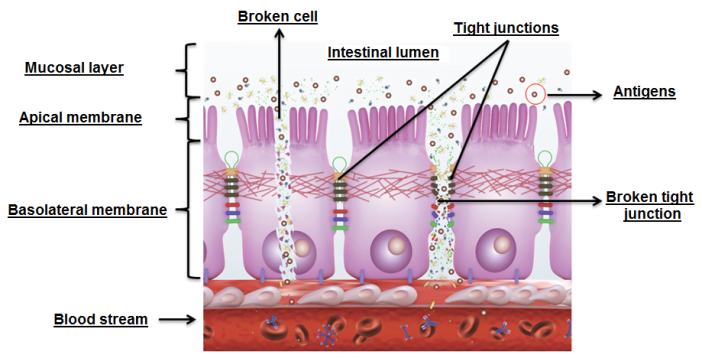
Figure 1. How antigens damage tight junctions.
These results can be explained by the quantification of circulating cells analyzed in the blood withdrew from these broilers. It is important to note that during a normal infection process, blood leukocytes are taken to the gut. However, if the animal has another type of infection, reduced total circulating leukocytes can impair response to the attack to this second antigen/site. This can be especially dangerous when total leukocyte blood levels are very low (leukopenia). In an analysis conducted in the study mentioned above, the group infected and supplemented with ImmunoWall® had reduced blood leukocyte mobilization to the gut at 14 days. However, when this immune system is subdivided, and different cells are analyzed, animals in this group have more APCs, suppressor monocytes (that prevent an uncontrolled immune response) and helper T lymphocytes (CD4 – they produce interleukins and stimulate the multiplication of cells that will attack the antigen) compared to the group of untreated challenged animals. The group of unchallenged animals given ImmunoWall® showed intermediate responses (between the challenged and the unchallenged control) to the analyzed cells mentioned above, in addition to cytotoxic T lymphocytes (CD8), which are important to prevent or control Salmonella invasion, as they can invade monocytes and then move to the liver and other organs.
Chart 1 below shows that supplementation with ImmunoWall® resulted in higher production of anti-Salmonella IgA at 14 days old. This indicates that the immune system had a quicker and stronger specific response, using less energy and nutrients, as the inflammatory response seemed to be shorter.
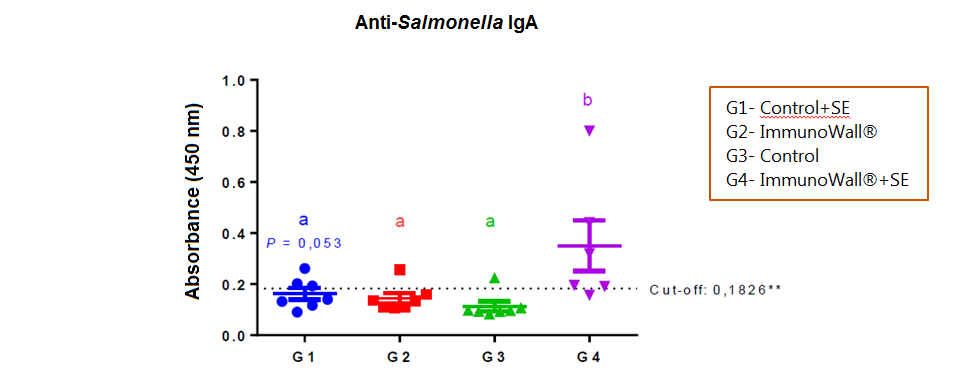
Chart 1. Relative IgA quantification in reagent serum against bacterial LPS. The cut-off line is shown. Different letters on each group indicate statistical relevance. ANOVA test with Tukey post-test (P<0.05, except when otherwise indicated).
SE can be a problem to broilers that still do not have a mature immune system, as they cannot fully control the infection. For this reason, most of the improved responses found in this study were observed up to day 14. Therefore, β-glucan supplementation can help broilers activate the immune system and have an early and quicker innate immune system response, reducing/minimizing the damages caused by pathogens and, consequently, performance impairment. This type of response is especially important in animals at early development stages, during reproduction, periods of stress and environmental challenges; acting as a prophylactic agent and increasing animal resistance, thus minimizing losses.
Several other studies have confirmed the efficacy of ImmunoWall® in reducing pathogen contamination in broilers and eggs (Hofacre et al., 2017; Ferreira et al., 2014), reducing mortality and improving productivity (Bonato et al., 2016; Rivera et al., 2018; Koiyama, et al. 2018), especially under challenge. There are no feed additives that can solve problems related to management, health plan, immunization, nutrition, water quality, among others. Additives are tools used to help control and prevent them. Intensive animal production is known to be extremely challenging. Therefore, strengthening the immune system can be one of the keys to higher productivity.
References
Beirão B. C. B. et al. Yeast cell wall immunomodulatory and intestinal integrity effects on broilers challenged with Salmonella Enteritidis. In: 2018 PSA Annual Meeting. San Antonio-Texas, USA. Proceedings…. 2018.
Bonato et al. Desempenho de frangos de corte alimentados com mananoligossacarídeos, parede celular de levedura e nucleotídeos de diferentes fontes. In: Conferência FACTA 2016 de Ciência e Tecnologia Avícolas, Atibaia. Proceedings….2016.
Dalloul, R. A., H. S. Lillihoj. Poultry coccidiosis: recent developments in control measures and vaccine development. Expert Rev. Vaccines, v. 5, p.143-163, 2006.
Ferreira, A.J.P. et al. Uso da associação de levedura e fonte de nucleotídeos na redução da colonização entérica por Salmonella Hiedelberg em frangos. In: Conferência FACTA 2014 de Ciência e Tecnologia Avícolas, Atibaia. Proceedings…. 2014.
Hofacre, C., et al. Use of a yeast cell wall product in commercial layer feed to reduce S.E. colonization. Proceedings of 66th Western Poultry Disease Conference, March 2017, Sacramento, CA, p. 76-78, 2017.
Koiyama, et al. Effect of yeast cell wall supplementation in laying hen feed on economic viability, egg production, and egg quality. The Journal of Applied Poultry Research, v. 27 (1), p. 116–123, 2018.
Lee, H. K., A. Iwasaki. Innate control of adaptive immunity: dendritic cells and beyond. Semin. Immunol., n. 19, p.48-55, 2007.
Petravić-Tominac, V. et al. Biological effects of yeast β-glucans. Agriculturae Conspectus Scientificus, n. 75, v. 4, 2010.
Rivera et al. Yeast cell wall and hydrolyzed yeast as a source of nucleotides effects on immunity, gut integrity, and performance of broilers. In: 2018 International Poultry Scientific Forum. Atlanta-GA, USA. Proceedings…. p. 49, 2018.
Rocha, T.M., Andrade, M.A., Santana, E.S., Fayad, A.R., Matias, T.D. Aspectos clínicos, patológicos e epidemiológicos de doenças imunossupressoras em aves. Enciclopédia Biosfera, Centro Científico Conhecer, v.10, n.18, p. 25, 2014.
Sharma, J. M. The avian immune system. In: Saif, Y. M. (ed.), Diseases of Poultry, 11th ed., pp. 5-16. Iowa State Press, Ames, IA, 2003.
Posted in 05 October of 2021


