Eubiotics in the modern production of broilers, concepts, and strategies of use

M.V. M.Sc. Dino Garcez – poultry consultant (DGbioss)
Eubiotics are nutritional additives that act essentially by improving the use of nutrients and maintaining intestinal health and, with this, promote poultry’s health, well-being, and performance. In addition, they can confer a greater innocuousness to the food. They are well-established tools in the world market of animal production, do not generate antimicrobial resistance (AMR) nor leave residues in the meat. Among the main eubiotic additives, we can mention: probiotics, prebiotics, symbiotics (pro + pre), organic acids, and phytogenic products.
This article aims to conceptualize these additives as to their composition, mode of action, main benefits, and to point out strategies of use in the modern production of broilers.
Probiotics
They are additives based on bacteria, yeasts, or fungi that alter the intestinal microbiota promoting a balance among the different beneficial bacterial populations that inhibit the development of pathogenic bacteria. As a result, they increase intestinal integrity, have an immunomodulatory response, which translates into improved zootechnical performance and contributes to greater safety of poultry products. The bacterial genera most used as probiotics are: Bacillus spp., Lactobacillus spp., and Enterococcus spp. In addition, yeasts of the species Saccharomyces cerevisiae are used in a concentrated manner.
The mode of action of each probiotic in the gastrointestinal tract (GIT) depends on the microorganism in question, below are listed the main mechanisms:
- Competition for adhesion sites and nutrients;
- Aggregation;
- Production of metabolites (organic acids and bacteriocins) that inhibit the development of pathogenic microorganisms (Clostridium perfringens, E. coli) and favor beneficial microbiota (e.g. Lactobacillus spp.)
- Synthesis of digestive enzymes and vitamins (e.g., B complex);
- Stimulation of antibody secretion (IgA) and activation of cells of the gut-associated lymphoid tissues (GALT);
- Reduction of ammonia concentration in the excreta;
In the initial phase of poultry’s life (0 to 7 days), there is a greater susceptibility to infection by pathogens in the intestine, largely due to the immaturity of the poultry immune system. Thus, probiotics are recommended from the first day of life, preferably in the hatchery, to limit the colonization of undesirable microorganisms (e.g. Salmonella spp.). However, probiotics are recommended not only in the first few days since numerous factors can alter the microbial population of the intestine such as: anti-nutritional factors (e.g. phytate), mycotoxins, coccidiosis challenges as well as management and biosecurity failures. Therefore, a continuous supply of probiotics via feed is recommended throughout the production cycle, especially in the early poultry life (0-21 days).
In selecting a probiotic additive, we must consider the viability of the microorganism(s) during storage, its resistance to the pelletizing processes of the feed (temperatures between 80°C to 90°C), and its survival and action in the GIT. In addition, it needs to be compatible with chemical anticoccidials and ionophores traditionally used in poultry farming.
Prebiotics
They are oligosaccharide-based additives such as MOS (mannan-oligosaccharides), FOS (fructooligosaccharides), GOS (galactooligosaccharides), and beta-glucans that have diversified effects on their host. These sugars are resistant to the digestive enzymes of poultry. However, they are used by beneficial bacteria populations of the GIT. Thus, they favor the beneficial microbiota (lactobacilli and bifidobacteria), reduce the population of pathogenic bacteria (E. coli and Campylobacter spp.), modulate the immune system, increase digestibility, absorption of minerals and vitamins, maintain intestinal pH and maximize the use of nutrients, which is reflected in increased productive performance of broilers (Scheme 1).
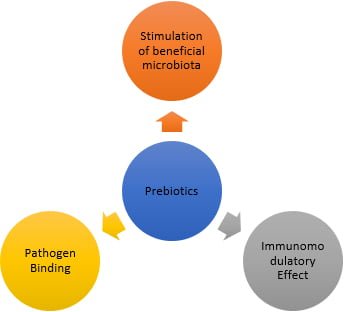
Scheme 1 – Main effects of prebiotics on the gut
The MOS have binding sites that bind to fimbriae receptors type 1 of Escherichia coli and Salmonella spp. which eliminates these bacteria from the GIT through the normal flow of the digesta. This phenomenon also implies an increase in the humoral immune response due to a greater presentation of antigens to the immune system. Beta-glucans (1.3 and 1.6), in turn, activate the immune system in a direct way because they bind to cellular receptors (dectin-1) present in enterocytes and phagocytes, and from there, they trigger a series of inflammatory reactions with emphasis on the activation of antibody (IgA) and mucus production, which are the first line of defense of the intestine against pathogens. In addition, beta-glucans activate phagocytes, natural killers, B and T lymphocytes,as increase cytokine production and phagocytic activity of macrophages. The immunomodulatory effect of MOS and B-glucans is summarized in regimen 2.
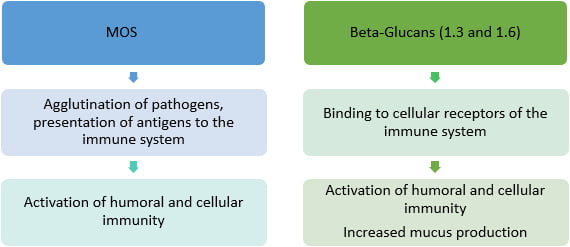
Scheme 2 – summary of the immunomodulatory mechanism of action of MOS and beta-glucans
Similar to probiotics, the use of prebiotics is indicated especially in the early stages of the poultry life and in conditions of high stress and infection pressure (e.g., high density).
In general, enzymatic processing (associated with optimal temperature, time and pH) reflects in prebiotics of higher efficiency than mechanically or chemically fractionated ones because it allows greater adhesion of the pathogen to the MOS due to the greater physical exposure of its molecule. Therefore, the processing is a relevant factor in the quality of the additive more than its concentration itself.
Innovatively, symbiotics (a combination of pro and prebiotics) have been developed as auxiliary tools in of preventing Salmonella spp. in poultry.
Organic acids (OAs)
They are composed of short-chain acids (one to seven carbons) with antimicrobial properties that relate to the reduction of the gastrointestinal tract (GIT) pH and its ability to dissociate its carboxyles (pKa value). When used in poultry, they can directly affecting the population of GIT bacteria, reducing pathogens and controlling, mainly, species that compete with poultry for nutrients, which results in improved productive performance of animals.
Table 1 below describes the main organic acids used in poultry farming.
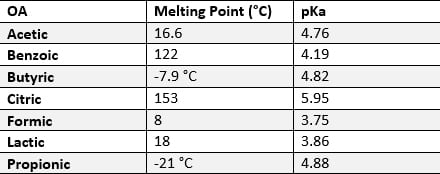
Table 1 – Main organic acids and physical properties
OAs in the undissociated form penetrate the bacteria’s cell wall and disrupt the normal physiology of certain “pH-sensitive” bacteria, which do not tolerate a wide internal and external pH gradient. Thus, OAs are more effective against “acid-intolerant” bacteria species such as E. coli, Salmonella spp., and Campylobacter spp., which results in less competition for nutrients with the host, reduced performance-depressant metabolites, and increased digestibility.
Its use is recommended throughout the production cycle, however, the initial stages of life (0-21 days) and the last week before slaughter are more strategic. In the first, some OAs (e.g., benzoic acid) prevent contamination by Salmonella spp. and increase digestibility by stimulating the activity of digestive enzymes (pepsin), which favors the zootechnical performance (weight gain and feed conversion) in this phase. On the other hand, its use in the withdrawal phase can contribute to reducing the microbial load of the poultry carried to the meat processing company, which implies greater food safety.
Phytogenic
They are additives composed of essential oils (EOs) and/or plant extracts (PEs) that have an antimicrobial (bacteria, viruses, and fungi), antioxidant, anti-inflammatory, and digestive effect by stimulating the production of digestive enzymes (e.g., amylase). The EOs are complex mixtures of volatile substances, usually lipophilic, whose components include terpenes, simple alcohols, aldehydes, ketones, phenols, esters, fixed organic acids, etc. When used in feed, they improve animal performance but do not have a drug effect, either by the active ingredient or its dosing.
Table 1 lists the main phytogenic additives and their respective primary modes of action.
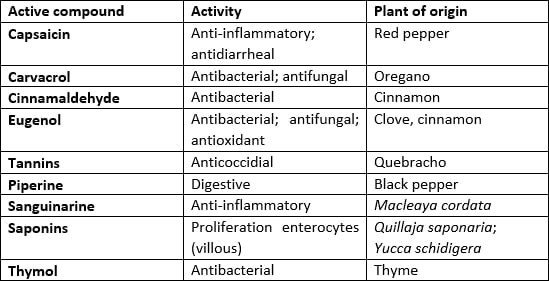
Table 1 – Phytogenic compounds, activity and origin
The use of phytogenic products in broilers can be employed throughout the production cycle, however, taking into account the limited production of endogenous enzymes (e.g., amylase) in the initial phase (0-21 d) as well as a higher prevalence of necrotic enteritis (C. perfringens) in this period, its use becomes strategic.
The stability of phytogenic additives is a crucial point in the quality of the product, so microencapsulation methods that reduce the volatility of the product “as-is” as well as protect against high temperature and pressure during pelleting of the feed are applied. Recently, gradual release technologies in GIT have increased the efficiency of EOs potentiating their effects on poultry.
The combined use of eubiotics should consider the main mode of action of the selected additives to avoid overlap (e.g., probiotic + probiotic). In addition, the compatibility between additives, as well as possible synergies, should be considered.
In a challenge study of Eimeria spp. and C. perfringens in broilers (0-28 d), the combination of prebiotic (MOS + beta-glucans) and essential oils (thymol + carvacrol) resulted in a zootechnical performance (weight gain, feed intake, feed conversion, and mortality) equal to the association of narasin and nicarbazin.
Table 2 shows the recommendation for use by phase and the best strategy for use (cost-benefit).
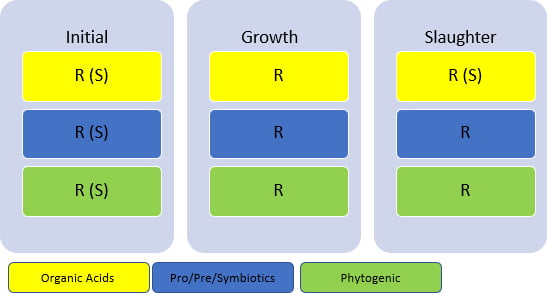
Table 2 – Recommendation for phased use (via feed) of eubiotics in poultry*
R = recommended, S = strategic. *May vary depending on the additive used and the manufacturer’s recommendation.
The supply of eubiotics has been significantly expanded from the recent restrictions on the use of AMDs worldwide . However, it is necessary to have criteria for the selection of these additives as well as the definition of a strategy for use to extract maximum efficiency from them without burdening the costs of poultry production. Currently, these additives are safely used in large poultry integrations because they have consistent productive benefits based on scientific data and contribute to sustainable food production.
Bibliographic References
AL-SAGAN, A. A.; ABUDABOS, A. M. Effect of eubiotic administration to broiler’s feed on intestinal morphology and microbiology under Clostridium perfringens challenge. Indian Journal of Animal Research, n. OF, 11 jun. 2016.
BAFUNDO, K. W.; MÄNNER, K.; DUERR, I. The combination of quillaja and yucca saponins in broilers: effects on performance, nutrient digestibility and ileal morphometrics. British Poultry Science, v. 62, n. 4, p. 589–595, 4 jul. 2021.
BARBALHO, R. L. DO C. et al. Β-glucans and MOS, essential oil, and probiotics in diets of broilers challenged with Eimeria spp. and Clostridium perfringens. Poultry Science, v. 102, n. 4, p. 102541, abr. 2023.
BURT, S. Essential oils: their antibacterial properties and potential applications in foods—a review. International Journal of Food Microbiology, v. 94, n. 3, p. 223–253, ago. 2004.
FASCINA, V. B. et al. Phytogenic additives and organic acids in broiler chicken diets. Revista Brasileira de Zootecnia, v. 41, n. 10, p. 2189–2197, out. 2012.
FULLER, R. Probiotics in man and animals. The Journal of Applied Bacteriology, v. 66, n. 5, p. 365–378, maio 1989.
GIRGIS, G. et al. Effects of a synbiotic combination of Bacillus subtilis and yeast cell wall-derived glucomannan on cecal colonization of Salmonella Enteritidis in layer chickens. Journal of Applied Poultry Research, v. 31, n. 2, p. 100240, jun. 2022.
GUIDOTTI, M.; CAFÉ, M. B. Aditivos fitogênicos na alimentação de aves de produção. 1. ed. [s.l.] Navegando Publicações, 2016.
KHADEM, A. et al. Growth promotion in broilers by both oxytetracycline and Macleaya cordata extract is based on their anti-inflammatory properties. British Journal of Nutrition, v. 112, n. 7, p. 1110–1118, 14 out. 2014.
KHAN, S. H.; IQBAL, J. Recent advances in the role of organic acids in poultry nutrition. Journal of Applied Animal Research, v. 44, n. 1, p. 359–369, jan. 2016.
LANGHOUT, P. Conferência Apinco de Ciência e Tecnologia Avícolas. Santos: Apinco. 2005.
ORNDORFF, B. W. et al. Comparison of Prophylactic or Therapeutic Dietary Administration of Capsaicin for Reduction of Salmonella in Broiler Chickens. Avian Diseases, v. 49, n. 4, p. 527–533, dez. 2005.
SANTURIO, J. M. et al. Atividade antimicrobiana dos óleos essenciais de orégano, tomilho e canela frente a sorovares de Salmonella enterica de origem avícola. Ciência Rural, v. 37, n. 3, p. 803–808, jun. 2007.
TONDA, R. M. et al. Effects of tannic acid extract on performance and intestinal health of broiler chickens following coccidiosis vaccination and/or a mixed-species Eimeria challenge. Poultry Science, v. 97, n. 9, p. 3031–3042, set. 2018.
WEBER, G. M. et al. Effects of a blend of essential oil compounds and benzoic acid on performance of broiler chickens as revealed by a meta-analysis of 4 growth trials in various locations. Poultry Science, v. 91, n. 11, p. 2820–2828, nov. 2012.
WENK, C. Herbs and Botanicals as Feed Additives in Monogastric Animals. Asian-Australasian Journal of Animal Sciences, v. 16, n. 2, p. 282–289, 1 jan. 2003.
Posted in 25 July of 2023


